What is Methyl Ethyl Ketone?
Methyl ethyl ketone (MEK) or butanone is a clear, volatile liquid with the formula C4H8O that is soluble in water and has a mild, acetone odour. The compound is biosynthesized by trees and is found in small quantities in foods such as some fruits and vegetables, chicken, beans, honey, and a wide variety of cheeses.
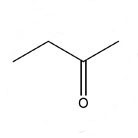
Technical Properties
Technical properties of Methyl Ethyl Ketone:
Cas Number: 78-93-3
Molecular Formula: C4H8O or CH3COCH2CH3
Synonyms: MEK, butanone, 2-butanone, butan-2- one, oxobutane, or methyl acetone
Molecular Mass: 72.11 g/mol
Exact Mass: 72.057514874 g/mol
Flashpoint (closed cup): 16 °F / -9 °C
Autoignition temperature: 961 °F / 516.11 °C
Boiling Point: 175 °F / 79.4 °C at 760 mm Hg
Melting Point: -123.3 °F / -86.28 °C
Vapour Pressure: 77.5 mm Hg at 68 °F / 20 °C
Density: 0.806 at 68 °F
Log P: 0.29
How is Methyl Ethyl Ketone produced?
The main commercial production route for methyl ethyl ketone products is through the dehydrogenation of secondary butanol although newer technology involves the direct oxidation of nbutene in solution using palladium and cupric chlorides as catalysts.
The Chinese market is the main driver for the demand of MEK and is also where several new productions plants have been opened in the last 5 years. World production now stands at approximately 320,000 to 350,000 tonnes.
How is Methyl Ethyl Ketone Stored and Distributed?
Methyl ethyl ketone is packed in drums. It should be stored in a cool, dry, well-ventilated area that is free from the risk of ignition as it is highly flammable. For transportation purposes, methyl ethyl ketone is classified as packing group II and a hazard class 3 with a flashpoint of -9° C (closed cup) and a specific gravity of 0.81. It is also an irritant.
Health Hazards
The compound is rated a 1 for health under the NFPA 704 indicating that exposure should only cause a minor residual injury.
To avoid inhalation occurring, especially for regular uses, adequate ventilation should be ensured with local exhaust ventilation and enclosures. The direct inhalation of high levels of MEK may cause dizziness, nausea, headaches, irritation of the respiratory system and chest tightness. If the individual has difficulty breathing, they should be moved to air free of the vapours and be provided oxygen until medical assistance is available.
Direct eye exposure can cause sore, stinging, red eyes with tearing and vapours can also irritate the eyes. Chemical safety goggles should therefore be used when contact with the eyes is possible. Repeated or prolonged skin contact should be avoided as this can cause irritation and dryness. Therefore, gloves, aprons and appropriate boots should be worn when repeated contact with MEK is possible.
Methyl Ethyl Ketone Uses
Solvent
The primary use of MEK is as a solvent with over 50% of demand coming from the paints and coating industry. The compound is used to enable a low viscosity solution that does not impact the film properties of the product.
These lacquers are used in the automotive, electrical goods and furniture industries. It is very effective as a surface coating solvent that it has become almost vital to the development of high solids coatings that reduce external emissions.
Plastic Wielding Agent
MEK is also commonly used in scale model sets, where a strong, durable bond is required between different model parts. When applied to a part, the surface plastic is dissolved and can then be wielded together by hand. The methyl ethyl ketone then evaporates away, leaving no residue to impact the appearance of the model and strong bonds that will be difficult to break apart.
Industry Uses
The compound can also be used in the manufacture of plastics and textiles, the manufacture of printing inks, adhesives, pesticides and in rubber-based industrial cements. It is used in the chemical industry as it is a precursor to methyl ethyl ketone peroxide which is itself used as a catalyst to initiate the polymerisation of polyester resins used in fibreglass reinforced products.
Solventis as your Methyl Ethyl Ketone Supplier
Solventis is a leading bulk supplier and distributor of methyl ethyl ketone or MEK in the UK, Europe and globally. As a company with offices in the UK and Belgium, we are proud to be able to offer a personal and efficient service, together with highly competitive prices.
As a result of our state-of-the-art facility in Antwerp, Belgium, we can guarantee the quality, efficiency, safety, and environmental awareness for our whole range of products that is second to none. Please contact us for more information on our methyl ethyl ketone or any ketone from our range on our enquiry form or by phone on +44 (0)1483 203224.



